This summary outlines the key points regarding the transition from the E2B(R2) to the E2B(R3) standard for Individual Case Safety Reports (ICSR) submissions.
Overview of ICSR E2B R2 vs. R3
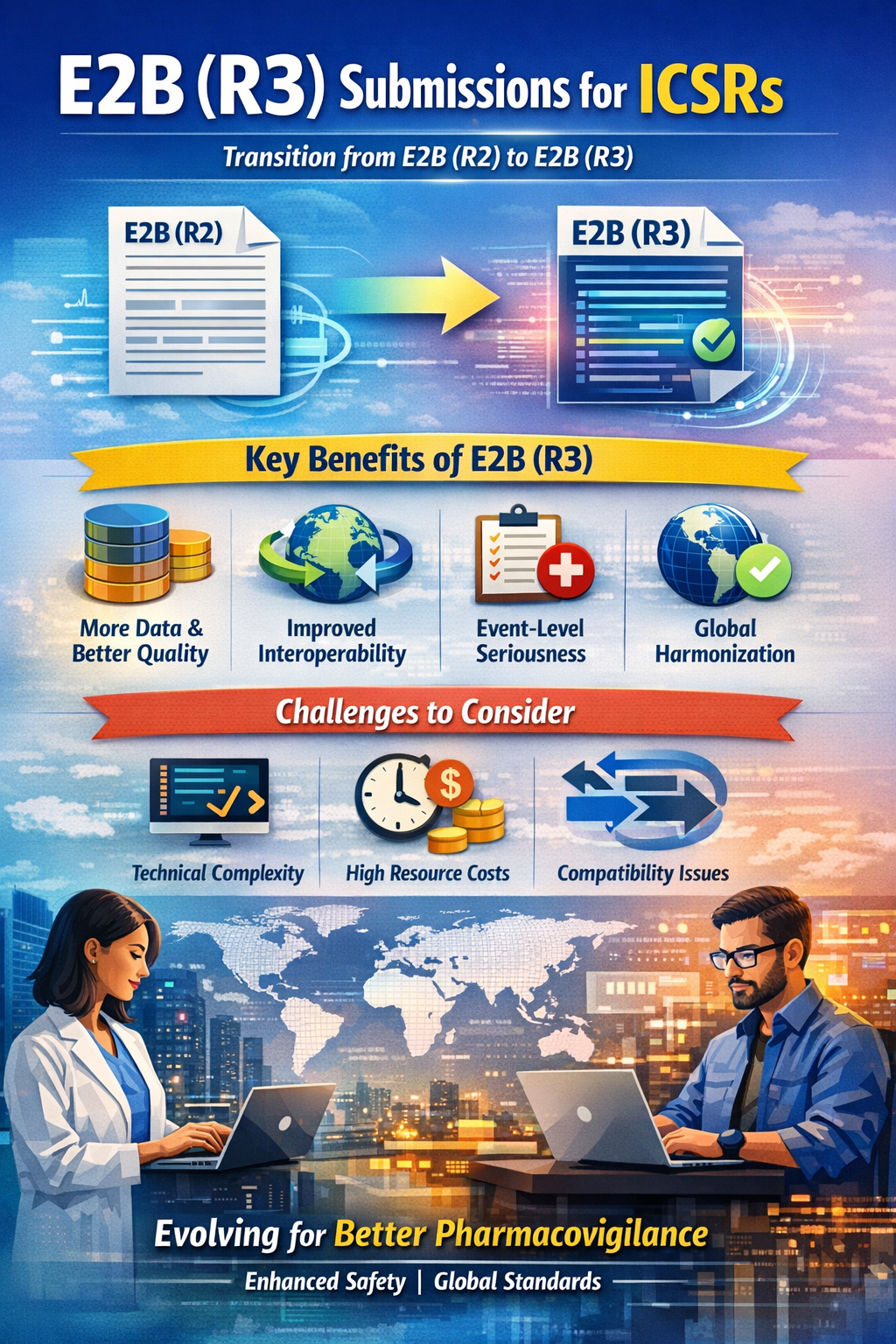
The E2B guideline, established by the International Council for Harmonization (ICH), provides the data elements and message specifications for the electronic transmission of ICSRs. While E2B(R2) was the industry standard for over a decade, the shift to E2B(R3) marks a move toward a more sophisticated, ISO-compliant (ISO 27953-2), and XML-based (HL7) framework.
Advantages of E2B(R3)
- Enhanced Data Quality and Granularity: R3 introduces a significantly higher number of data elements (increasing from approximately 271 in R2 to over 330 in R3). This allows for more structured data rather than relying on free-text comments, leading to better data mining and signal detection.
- Interoperability: Because R3 is built on HL7 standards, it facilitates seamless data exchange between different healthcare systems, clinical trial databases, and regulatory authorities.
- Improved Seriousness Assessment: In R2, seriousness was often assessed at the case level. R3 allows for seriousness to be recorded at the event level, providing a clearer clinical picture when a patient experiences multiple reactions. This is an impactful change for Medical reviewers and beneficial for signal management.
- Global Harmonization: R3 aligns more closely with international standards (IDMP – Identification of Medicinal Products), reducing the need for region-specific "tweaks" and supporting a more unified global safety database.
- Efficiency in Follow-ups: The structured nature of R3 makes it easier to track changes and updates to a case, as specific elements can be updated without re-submitting or re-processing the entire narrative from scratch.
Limitations and Challenges
- Technical Complexity: The transition from R2 to R3 requires a complete overhaul of pharmacovigilance (PV) software. The XML structure of R3 is much more complex than the SGM-based R2, requiring specialized IT expertise for implementation.
- Resource Intensity: Training staff on the new data fields and the logic of R3 is time-consuming. For smaller companies, the cost of upgrading systems and validating new processes can be a significant financial burden.
- Backwards/Forwards Compatibility (BFC): During the transition period where some regions use R2 and others use R3, companies must use conversion tools. These conversions can sometimes lead to data truncation or "loss in translation," where R3-specific details are lost when converted back to the simpler R2 format.
- Increased Data Entry Time: With more mandatory and highly recommended fields, the initial processing time for a single ICSR may increase, potentially straining safety teams during peak reporting periods (e.g., during a pandemic or a major product launch).
Conclusion
The transition to E2B(R3) is a necessary evolution for modern pharmacovigilance. While the limitations are primarily technical and operational hurdles during the implementation phase, the long-term advantages—specifically in terms of patient safety, better data analytics, and global regulatory alignment—far outweigh the initial costs. Organizations must focus on robust data migration strategies and staff training to successfully bridge the gap between these two standards.
To learn more from related topics, please visit our website or newsletter at https://medipharmsolutions.com/newsletter/

No Comments